III. Compound theory
Key focus of this chapter: naming chemical compounds
This chapter focuses on naming chemical compounds and gives concise summaries of the important things about covalent bond which contains electronegativity, polar and nonpolar bond, dipole moments of polar and nonpolar molecules, molecular forces, octet rule, VSEPR-Valence shell, formal charges, and hybridization in more detail.
A. Naming chemical compounds
1. Naming oxoanions (atom + oxygen(s))
Number of oxygen atom(s) | Prefix | Suffix | Examples |
One | Hypo - | - ite | |
Two | - ite | ||
Three | - ate | ||
Four | Per - | - ate |
2. Naming acids
a. Oxoacids (hydrogen + oxoanion)
Number of oxygen atom(s) | Prefix | Suffix | Examples |
One | Hypo - | - ous acid | |
Two | - ous acid | ||
Three | - ic acid | ||
Four | Per - | - ic acid |
** H2SO3 : Sulfurous acid
H2SO4 : Sulfuric acid
H3PO4 : Phosphoric acid
b. Hydrogen + anion
Prefix | Suffix | Examples |
Hydro - | - ic acid | HCl(aq): hydrochloric acid HBr(aq): hydrobromic acid HI(aq): hydroiodic acid HCN(aq): hydrocyanic acid |
3. Table of naming polyatomic anions and acids
Poly atomic anions | Acids | |||
Single charged anions | Hydrogen carbonate (bicarbonate) | Carbonic acid | ||
Hydrogen sulfate (bisulfate) | Sulfuric acid | |||
Dihydrogen phosphate | Phosphoric acid | |||
Acetate | Acetic acid | |||
Cyanide | Hydrocyanic acid | |||
Nitrite | Nitrous acid | |||
Nitrate | Nitric acid | |||
Hydroxide | ||||
Hypochlorite | Hypochlorous acid | |||
Chlorite | Chlorous acid | |||
Chlorate | Chloric acid | |||
Perchlorate | Perchloric acid | |||
Permanganate | Permanganic acid | |||
Hypobromite | Hypobromous acid | |||
Bromite | Bromous acid | |||
Bromate | Bromic acid | |||
Perbromate | Perbromic acid | |||
Double charged anions | Hydrogen phosphate | Phosphoric acid | ||
Carbonate | Carbonic acid | |||
Chromate | Chromic acid | |||
Dichromate | Dichromic acid | |||
Peroxide | Hydrogen peroxide | |||
Sulfite | Sulfurous acid | |||
Sulfate | Sulfuric acid | |||
Thiosulfate | Thiosulfuric acid | |||
Triple charged anions | Phosphate | Phosphoric acid | ||
** NH4+: Ammonium
4. Naming compounds
Fig. 1 Classification of groups

Combinations | Naming compounds | |
 | ||
 |  | |
 | ||
 |  | |
 |  | |
B. Chemical Bonds
Chemical Bonds | Features |
Ionic bonds |  |
Metallic bonds |  |
Covalent bonds (molecules) |  |
C. Covalent bond
1. Electronegativity (EN)
- Atomic force to pull apart the shared electrons in a covalent bond.
- The atoms of higher electronegativity have strong nonmetallic character, and they easily become anions.
- A greater difference of the electronegativity between two atoms means a higher polarity.
- In the same period, electronegativity increases as the atomic number increases.
- In the same group, electronegativity decreases as the atomic number increases.
Fig. 2 Electronegativity in the periodic table

2. Polar and nonpolar covalent bond
Classification | Features |
Polar covalent bond | • The bigger difference of electronegativity between two atoms in molecules, the bigger polarity of the molecules • Big difference of electronegativity between two atoms → the higher polar molecule → ionic bonds |
Nonpolar covalent bond | • The difference of electronegativity between diatomic elements is zero (H2 ,F2, Cl2) • Usually C-H bond is considered to be nonpolar. |
- Ex/ The order of polarity between two atoms:
 |
Therefore, ionic bonds (NaF, LiF) have a higher polarity than covalent bonds (HF, CH, F2)
3. Dipole moments of polar and nonpolar molecules
Classification | Features |
Polar molecules |  |
Nonpolar molecules |  |
4. Molecular forces (van der Waals force)
: attractive forces between molecules
Classification | Features |
London dispersion force |  |
Dipole-dipole force |  |
Hydrogen bond |  |
Ion-dipole |
** Order of strength between molecules when they have a similar molecular weight
London dispersion force < Dipole – dipole force < Hydrogen bond
 |
** The boiling and melting point of molecules are usually dependent on London dispersion force (molecular size or weight) rather than dipole-dipole force.
– the strength order of dipole-dipole force : HI < HBr < HCl
– the strength order of London dispersion force: HCl < HBr < HI
– the strength order of the boiling and melting point: HCl < HBr < HI
** Other covalent bonds
Diatomic molecules |  |
Coordinate covalent |  |
Covalent network solid |  |
5. Octet rule
: atoms in the main group tend to be positioned with 8 electrons in their outmost shell like noble gases.
a. Valence electron:
- Electrons of atoms in the outermost shell
- Forming covalent bonds
- Main factor to distinguish the chemical properties of atoms
- Ex/
Atoms | Number of valence electrons | Forming bonds |
C N O F | 4 5 6 7 | 4 3 2 1 |
b. Lewis structure
- Repulsive and attractive forces make optimum distance between two atoms
– repulsive forces between two different atom’s electrons
– attractive forces between one atom’s electrons and another atom’s nucleus
- Indicating covalent bonds
– double bonds by sharing 4 electrons (2 pairs): CO2, O2
– triple bonds by sharing 6 electrons (3 pairs): HCN, N2
c. Exception to octet rule
- Some atoms (Be, B, Al) have less than 8 electrons in their outermost shell.
- Some atoms (past the second row in the periodic table: Si, P, Ge, Sn,…), have more than 8 electrons in their outermost shell.
Fig. 3 atoms that are exceptions to the octet rule

- By using low energy in d orbitals the atoms could expand their valence shell to fill out more than 8 electrons
 |
6. VSEPR-Valence shell (valence-shell electron-pair repulsion model)
: electrons will be positioned as far as possible from one another to maintain a stable state of molecular geometry by the repulsive forces.
Molecular shapes | Molecular geometry | Examples | Features |
Linear |  |  |  |
 |  |  | |
Trigonal planar |  |  |  |
Bent |  | 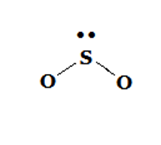 | 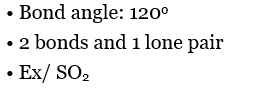 |
 |  |  | |
Tetrahedral |  |  |  |
Trigonal pyramidal |  |  |  |
Trigonal bipyramidal |  |  |  |
Seesaw |  |  |  |
T-shaped |  | 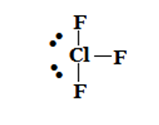 |  |
Octahedral |  |  |  |
Square pyramidal |  |  | 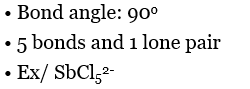 |
Square planar |  |  |  |
7. Formal charges
: atomic charges in a molecule.
 |
 |
Atoms | Number of valence electrons | Binding number | Number of non-bonding electrons |
N | 5 | 3 | 2 |
C | 4 | 4 | 0 |
S | 6 | 1 | 6 |
Formal charge of N: 5 −3 − 2 = 0
Formal charge of C: 4 − 4 − 0 = 0
Formal charge of S: 6 − 1 − 6 = -1
8. Hybridization
: new orbitals by overlapping atomic orbitals.
a. Sigma(σ) bond
- Axis bond by head-on orbital overlap
- Straight orbital overlap
- Round shape by vertical section
- Occupying maximum 2 electrons
b. Pi(π) bond
- Sideways overlap
- Dumbbell-shape by vertical section
- Weaker than sigma(σ) bond
c. Application
- Single bond: 1 sigma bond and 0 pi bond
- Double bond: 1 sigma bond and 1 pi bond
- Triple bond: 1 sigma bond and 2 pi bonds
d. Hybrid orbitals
Classification | Total number of bonds & lone pairs | Shape of molecules | Examples |
• Linear | |||
• Bent • Trigonal planar |  | ||
• Bent • Trigonal pyramidal • Tetrahedral | 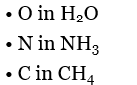 | ||
• Linear • Seesaw • T-Shaped • Trigonal bipyramidal |  | ||
• Octahedral • Square Pyramidal • Square Planar |  |
- Ex/
BF3 : 3 bonds and 0 lone pair → 3+0=3 → sp2 types of orbital
NH3: 3 bonds and 1 lone pair → 3+1=4 → sp3 types of orbital
9. Resonance
: molecule structures, which are not represented by Lewis structure, show two or more valence-bond structures by moving their electrons.
- More resonance structures, greater stabilization
- Maintaining same number of unpaired electrons
- Ex/ O3, OCN–, NO3–
D. Magnetism
Magnetism | Features |
Ferromagnetism |  |
Paramagnetism |  |
Diamagnetism |  |
E. Unit cells
: the basic unit of crystal structure arranged by atoms is expressed by lattice, which is the center of an atom with 3-D repeated array.
Unit Cell | Number of atom(s) per unit cell | Features |
Primitive cubic (Simple cubic) | 1 | • 1/8 of each atom in unit cell • Ex/ Polonium |
Body-centered cubic | 2 | • 1 atom at center with 1/8 of each corner atom • Ex/ Tungsten, iron, sodium |
Face-centered cubic | 4 | • 1/2 of each face-centered atoms with 1/8 of each corner atom • Ex/ Copper, silver, gold, nickel, lead, diamond |
