II. Orbital and Periodicity questions

Electron configuration questions
There are a total of 43 orbital, periodicity, and clectron configuration questions, which are important content-oriented in the summary part of this chapter. Click the ‘Answer’ button to see the corresponding answers and explanations, and click the ‘Go back’ button to slove the next questions.
1. Choose the INCORRECT statement about orbital theory.
A. Modern atomic model indicates a high probability of finding certain positioned electrons around the nucleus.
2. The orbital is the path of an electron circling around a nucleus. Unlike a planetary orbit, this path is not fixed.
3. When an electron is transferred from low to high shell, energy is absorbed.
4. The paths of electrons circling around nucleus of a hydrogen atom are described by Bohr’s atomic model.
5. All are correct.
2. When energized “excited” electrons in the outer shell fall down to the n=2 shell, energy is emitted. Which of the following is true for that statement?
A. Lyman series
B. Balmer series
C. Paschen series
D. Infrared region
E. C and D
3. Which of the following movements of an excited electron emits the highest energy?
A. N (n=4) → M (n=3)
B. N (n=4) → K (n=1)
C. N (n=4) → L (n=2)
D. K (n=1) → N (n=4)
E. L (n=2) → N (n=4)
4. Which of the following is the correct statement about the shapes of orbitals?
A. Dumbbell-shaped p orbitals exist at any electron shell.
B. Spherical shaped s orbitals have three orientations of coordinate axes, Px, Py, or Pz.
C. d orbitals have cloverleaf shapes and appear in the non-metal region of the periodic table.
D. f orbitals have 7 different orbitals and contain 7 electrons total in the orbitals.
E. The electron shell n=3 contains s, p, and d orbitals.
5. Which of the following does NOT describe how electrons fill orbitals?
A. Each orbital can hold one or two electrons.
B. The 3d orbital has a higher energy level than the 4s orbital.
C. Electrons in each orbital spin in opposite directions.
D. The electrons are filled into orbitals from high energy level to low energy level.
E. Electrons are half-filled into each orbital and then fully filled.
6. Which of the following is the most important indicator of the energy level of an orbital?
A. Principal quantum number (n)
B. Angular momentum quantum number (l)
C. Magnetic quantum number (ml)
D. 3-D shape of the orbital
E. Spin quantum number (ms)
7. Which of the following is a possible configuration for quantum numbers?
A. n=1, l=1, ml =0
B. n=2, l=1, ml =+2
C. n=2, l=3, ml =+3
D. n=3, l=2, ml =-2
E. n=4, l=4, ml =-1
8. One shell can hold a maximum of 32 electrons. This principal quantum number is _______________.
A. n=1
B. n=2
C. n=3
D. n=4
E. n=5
9. Which of the following is a possible quantum number when orbital notation is 4d?
A. n=2, l=1
B. n=3, l=3
C. n=3, l=2
D. n=4, l=3
E. n=4, l=2
10. Which of the following could be the lowest principal quantum number to be involved in a d orbital?
A. 1
B. 2
C. 3
D. 4
E. 5
11. Which of the following is an INCORRECT statement?
A. The total number of orbitals in the second shell is 3.
B. The third shell (n=3) can hold maximum 18electrons.
C. The principal quantum number of n=4 has four types of orbitals, s, p, d and f.
D. Angular-momentum quantum number, l, expresses the 3-D shape of the orbital.
E. The magnetic quantum number, Ml, expresses the spatial orientation of the orbital coordinate axes.
15. Which of the following pairs has the same number of unpaired electrons in orbitals when they are the ground state?
A. Boron, oxygen
B. Phosphorus, argon
C. Titanium, nickel
D. Sodium, magnesium
E. Titanium, cobalt
16. Which of the following is the INCORRECT statement about the electron configuration of [Ar]4s23d1?
A. This atom is Scandium.
B. [Ar]4s13d2 could be said to be the excited state of this atom.
C. There are 8 electrons at l=0 of angular momentum quantum number.
D. There is only 1 unpaired electron.
E. There is only 1 outermost electron.
17. How many orbitals for silicon are filled by electrons at ground state?
A. 7
B. 8
C. 9
D. 14
E. 16
21. Which of the following radiations is the highest frequency of a photon?
A. Radio waves
B. Microwaves
C. Gamma rays
D. Ultraviolet light
E. Visible light
22. Which of the following is INCORRECT?
A. High energy has short wavelength.
B. High frequency has short wavelength.
C. Low energy has low frequency.
D. The wavelength of visible light is longer than radio waves.
E. Usually all types of light have the same speed.
23. Which of the following does NOT apply to metal?
A. Easy oxidized
B. Brittle
C. Ductile
D. Malleable
E. Characterized by luster and shine
24. Which of the following is true for atoms in the periodic table?
A. In the same group atoms have the same number of shells.
B. Alkali metal group has a high electro affinity.
C. Atoms in the same period have similar chemical properties.
D. The halogen group is a strong reducing agent.
E. Alkaline earth metals have two electrons in their outermost shell.
25. Which of the following is an INCORRECT example for the atom of each group?
A. Alkali metal = H
B. Transitional metal = Zn
C. Halogen = I
D. Noble gas = Kr
E. Alkali earth metal = Ba
26. Which of the following has chemical property similar to chlorine?
A. Sulfur
B. Oxygen
C. Fluorine
D. Argon
E. Magnesium
27. Which of the following has intermediate chemical properties between metals and nonmetals?
A. Ga
B. Al
C. Ge
D. Sn
E. Se
30. Which of the following has the largest atomic radius?
A. Nitrogen
B. Oxygen
C. Fluorine
D. Phosphorus
E. Sulfur
31. Which of the following is correct pair for the comparison of the atomic radius?
A. Rb+ < Rb, Br– < Br
B. Rb+ > Rb, Br– < Br
C. Rb+ > Rb, Br– > Br
D. Rb+ < Rb, Br– > Br
E. Rb+ = Rb, Br– = Br
32. There are five isoelectronics, O2-, F–, Ne, Na+, and Mg2+. Choose the correct order from smaller to larger atomic radius.
A. O2- < F–< Ne < Na+ < Mg2+
B. Mg2+< Na+< Ne< F–< O2-
C. Ne < F–< O2-< Mg2+< Na+
D. O2-<F–<Ne < Na+ < Mg2+
E. Ne = F–= O2-= Mg2+= Na+
33. Which of the following is the largest second ionization energy to get rid of its outmost electron?
A. [Ne]3s23p3
B. [Ne]3s23p5
C. [Ar]
D. [Ar]4s1
E. [Ar]4s2
36. Which of the following electro configurations has the lowest electron affinity?
A. 1s22s1
B. 1s22s22p1
C. 1s22s22p2
D. 1s22s22p3
E. 1s22s22p4
38. Of the following, which is the correct order for electron affinity from lower to higher?
A. Si < P < S < Cl
B. P < Si < S < Cl
C. P < S < Si < Cl
D. Si < Cl < P < S
E. Si < P < Cl < S
41. In the periodic table, the following is true going in the direction left and down.
A. London dispersion is increased.
B. Atomic radius is decreased.
C. Electronegativity is increased.
D. Metallic character is decreased.
E. Ionization energy is increased.
43. Which of the following is the INCORRECT statement?
A. Within a group, the metallic character is increased with the increasing atomic number.
B. The radius of natural metallic atoms is usually larger than its cationic radius.
C. In the same period the radius of atoms is decreased with the increasing atomic number.
D. In the same period the atomic density is increased with the increasing atomic number.
E. All are correct.
Answers
2. B
The Lyman series of the Ultraviolet region emit energy when excited electrons fall down to n=1 shell. The Balmer series of the Visible region emit energy when excited electrons fall down to n=2 shell. The Paschen series of the Infrared region emit energy when excited electrons fall down to the n=3 shell.
3. B
The difference of the energy level is K(n=1) < L(n=2) < M(n=3) < N(n=4). To be absorbed the strong energy and become an excited electron, the electron must move from the lowest shell K (n=1), near the nucleus, to higher shells. To emit strong energy, excited electrons must fall from any of the high shells back to the lowest shell K (n=1).
4. E
Dumbbell-shaped p orbitals exist at all electron shells except n=1. Spherical shaped s orbitals are spread near nucleus with no direction. Three orientations of coordinate axes, Px, Py, or Pz, are features of p orbitals. Cloverleaf-shaped d orbitals appear in the transition metal region of the periodic table. Each orbital holds a maximum of 2 electrons; therefore 7 different f orbitals contain 14 electrons.
5. D
The electrons are filled into orbitals from low energy level to high energy level. 1s<2s<2p<3s<3p<4s<3d<4d …
6. A
The principal quantum number, n, represents the position of shells, the number of orbital types, and the energy level of orbitals. The angular momentum quantum number, l, indicates a 3d shape of the orbital. The magnetic quantum number, ml, indicates orbital axes of spatial orientation. The spin quantum number, ms, indicates the direction of spins.
8. D
The principal quantum number of n=4 contains a total of 16 (1+3+5+7) orbitals. Each orbital can hold a maximum of 2 electrons. Therefore, 16 × 2 =32 maximum electrons can be held in n=4. (Maximum number of electrons in shell: 2n2 → 2n2 = 32, n=4)
10. C
The principal quantum number of n=1 has only s orbital and the principal quantum number of n=2 has s and p orbital. From the principal quantum number of n=3, d orbital begins to be involved.
12. A
From the lower to higher energy in orbitals:
1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p → 5s → 4d → 5p → ….
16. E
The electro configuration of Sc is 1s22s22p63s23p64s23d1. The orbitals, which are l=0 of angular momentum quantum number, are 1s, 2s, 3s, and 4s. Therefore, the total electrons in 1s, 2s, 3s, and 4s orbitals are 4 ×2 = 8. There are 2 outermost electrons contained in 4s orbital.
17. B
The ground state electro configuration of silicon is 1s22s22p63s23p2. 1s2 = 1 orbital, 2s2 = 1 orbital, 2p3 = 3 orbitals, 3s2 = 1 orbital, 3p2 containing 2 electrons = 2 orbitals. Therefore, the total number of orbitals filled by electrons is 1+1+3+1+2 = 8.
18. B
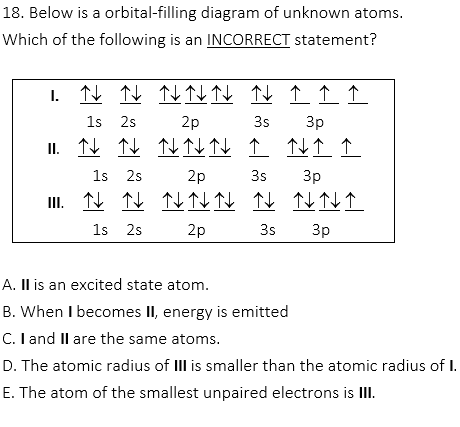
I is the ground state atom and II is the excited state of P atom. When the ground state atom becomes the excited state atom, energy is absorbed. III is Cl atom. Therefore, atomic radius is P > Cl. I and II have 3 unpaired electrons and III has 1 unpaired electron.
21. C
E = hν
The higher frequency (ν), the higher energy (E). The order of energy from lower to higher:
Radio waves < Microwaves < Infrared light < Visible light < Ultraviolet light< X rays < Gamma rays
23. B
Metal: easy oxidation (reducing agent), malleability, ductility, shiny luster, and good conductivity for heat and electricity.
Nonmetal: easy reduction (oxidizing agents), brittleness, and poor conductivity for heat and electricity.
24. E
In the same periods, atoms have the same number of shells. Halogen group has a higher electro affinity. In the same group, atoms have similar chemical properties because of the same outermost electrons (valence electrons). Alkali metal group is the strong reducing agents and halogen group is the strong oxidizing agents.
26. C
Atoms positioned in the same groups have similar chemical properties. Fluorine and chlorine belong to the same 7A group.
27. C
The elements that have intermediate chemical properties between metals and nonmetals, are called semimetal or metalloids. B, Si, Ge, As, Sb, Te, and At belong to this semimetal group. Ga, Al, and Sn are metals, and Se is a nonmetal.
28. B
Atoms of the 7A group are halogens, which are nonmetals (except astatine), acting as Lewis base when it is forming (–1) anions (**see the acid and base theory in Ch IX). Paramagnetic and forming cations are metals.
29. E
Transition metals, between 2A and 3A group, contain d orbitals and are paramagnetic. Calcium is belongs to the alkali earth metals. Transition metals form multiple oxidation states.
30. D
In the periodic table, going in the direction left and down, the radius of atoms becomes bigger. Phosphorus is positioned at the far left and down in the period table among the atoms.
31. D
The radius of a cation is smaller than its natural atom because the number of shells is decreased. The radius of an anion is bigger than its natural atom because the increasing repulsion between electrons causes an increase in the anionic radius.
32. B
The size of radius between isoelectronic elements: bigger cations < natural atoms < bigger anions
33. D
The atom with fully-filled electrons in orbitals has strong first ionization energy ([Ar]). The atom with 1 outermost electron has strong second ionization energy ([Ar]4s1). The atom with 2 outer most electrons has strong third ionization energy ([Ar]4s2).
34. B
In the same period with the increasing atomic number, the Ei is increased. In the same group with the increasing atomic number, the Ei is decreased. The half-filled or fully-filled electron(s) in each orbitals have a strong Ei, such as N or Ne. → N > O
35. B
The chart shows the high gap of ionization energy between E2 and E3, indicating that this group has strong E3 (third ionization energy). Atoms containing 2 outmost electrons (alkali earth metals) have strong E3.
36. D
In the same period, with increasing atomic numbers, the Eea is increased. An atom that emits one electron from half-filled or fully-filled orbitals has a strong Eea such as 1s22s22p2 or 1s22s22p5, but an atom that has half-filled or fully-filled electron(s) at orbitals has a weak Eea such as 1s22s22p3 or 1s22s22p6.
37. C
Electron affinitive (Eea) means negative (emitting) energy when an atom accepts an electron and become an anionic atom. The fluorine atom, F, has 7 electrons at the outermost orbital. This atom has the strong affinity to accept 1 electron for becoming a stable ionic atom, which will have 8 electrons at the outermost orbital.
38. B
In the same period, with the increasing atomic number, the Eea is increased. In the same group, with the increasing atomic number, the Eea is decreased. An atom that emits one electron from half-filled or full-filled orbitals, have a strong Eea such as Si or Cl. P < Si
39. A
The halogen group usually has strong electron affinitive because the atoms of 7 outermost electrons want to attract 1 electron to be in a stable state. Going in the direction left and down on the periodic table, the atomic radius becomes bigger. Noble gases have strong first ionization energy because of the stable 8 outermost electrons. The attraction between nucleus (+ charge) and electrons (-charge) is increased with the close distance between them. Therefore, He has higher first ionization energy than Ne.
40. E
Electronegativity is decreased going in the direction left and down in the period table.
41. A
London dispersion is the forces of electron motions in atoms or molecules. London dispersion forces increase with the size of atoms or molecules. Atomic size and London dispersion forces are increased when going in the direction left and down in the periodic table.
42. C
Atomic radius, London dispersion force, and metallic character are increased in the direction left and down in the period table. In the same direction, ionization energy, electro affinity, and electronegative are decreased.
43. E
All are correct statements. In the same period the atomic radius is decreased and atomic density is increased with the increasing atomic number because increasing number of protons and electrons causes an increase in the attraction between themselves.