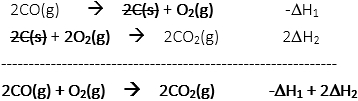V. Thermochemistry questions

Thermochemistry practice problems
There are a total of 11 phase change problems, which are important content-oriented in the summary part of this chapter. Click the ‘Answer’ button to see the corresponding answers and explanations, and click the ‘Go back’ button to slove the next questions.
1. When 75J of work is done on system, the ΔE is 100J. Which of the following is the correct statement about heat energy?
A. 25J of heat is absorbed into the system from its surroundings.
B. 25J of heat is released into the surroundings by the system.
C. 175J of heat is absorbed into the system from its surroundings.
D. 175J of heat is released into the surroundings by the system.
E. There is no change of the heat energy between system and its surroundings.
2. A cylinder is compressed from 5L to 3L volume by the pressure of 4 atm. How much work is done?
A. +8 atm·L
B. –8 atm·L
C. +12 atm·L
D. –12 atm·L
E. +20 atm·L
3. What is the work when water in a cylinder is evaporated and expanded from 10L to 15L against an external pressure of 3 atm?
A. + 1.67 atm·L
B. – 1.67 atm·L
C. +15 atm·L
D. –15 atm·L
E. – 45 atm·L
4. What is the mass of water when 420J of heat is used to raise the temperature from 30.0oC to 35.0oC? (Specific heat of water: 4.2 J/(g•oC))
A. 5.0g
B. 10.0g
C. 15.0g
D. 20.0g
E. 25.0g
5. How much energy is required to raise the temperature of 45g of NaCl from 35.0oC to 60.0oC? (Specific heat of NaCl: 0.9 J/(g•oC))
A. 135.0 J
B. 510.5 J
C. 1012.5 J
D. 1417.5 J
E. 2430.0 J
6. Which of the following applies the second law of thermodynamics?
A. Formation of liquid water from steam.
B. Producing sugar in a cell.
C. The sum of total energy from the system and its surroundings is unchanged.
D. Dissolution of NaCl in water.
E. Non-spontaneous process.
7. Which of the following applies to the positive change of enthalpy, +ΔH?
A. System gives off heat energy to its surroundings.
B. Reactants have higher energy than their products.
C. The heat of the surroundings is decreased.
D. The total energy is increased.
E. System becomes exothermic.
8. Which of the following are NOT examples of exothermic processes?
A. Combustion of propane
B. Acid and base neutralization
C. Cold pack by chemical reaction of ammonium nitrate in water
D. Catabolism of glucose in a cell
E. Making ice from evaporated water
9. The combustion of acetylene, C2H2, with oxygen is gives off 1,255 kJ of heat energy in the following equation. How much heat is given off by the reaction of 2 moles of acetylene?
C2H2(g) + 5/2 O2 (g) → H2O(g) + 2CO2(g) ΔHo = -1,255 kJ
A. 627 kJ
B. 1,255 kJ
C. 2,510 kJ
D. 3,137 kJ
E. 3,525 kJ
10. How many grams of NH3 is used when NH3 is reacted with excess O2 and 150kJ of heat is released in the following reaction?
4NH3(g) + O2(g) → 2N2H4(l) + 2H2O(l) ΔH = – 300 kJ
A. 4.3g
B. 8.5g
C. 17.0g
D. 34.0g
E. 68.0g
16. Which of the following represents the greatest increasing entropy for the compound AB?
A. AB(s) → AB(g)
B. AB(g) → AB(g)
C. AB(g) → AB(l)
D. AB(g) → AB(s)
E. AB(s) → AB(l)
17. When a solute is dissolved in a solvent, the temperature of its system is increased. Which of the following applies to its surroundings?
A. ΔT<0, ΔS<0
B. ΔT>0, ΔS<0
C. ΔT<0, ΔS>0
D. ΔT>0, ΔS>0
E. ΔT=0, ΔS=0
20. What temperature is needed for a spontaneous reaction when ΔH is 5 kJ/mol and ΔS is 20 J/mol•K?
A. 230K
B. 235K
C. 240K
D. 245K
E. 255K
Answers
1. A
ΔE = W + Q
Work is done on system → W = + 75J
100J = (+75J) + Q
Q = +25J
Therefore, 25J of heat is absorbed into the system from its surroundings.
6. D
The second law of thermodynamics means the law of disorder with spontaneous processes. A and B are non-spontaneous processes. C is the first law of thermodynamics.
7. C
• System gives off energy to its surroundings. • System becomes exothermic. • The heat of the surroundings is increased. • Reactants have higher energy than products. • Total energy is not changed. • Ex/ State change: gas → liquid → solid | • System absorbs energy from its surroundings. • System becomes endothermic. • The heat of the surroundings is decreased. • Reactants have less energy than products. • Total energy is not changed. • Ex/ State change: solid → liquid → gas |
8. C
All are examples of exothermic processes except C. Because the system gains heat from its surroundings, the temperature of the surroundings is decreased. The cold pack by chemical reaction of ammonium nitrate in water is an example of an endothermic process.
13. B
ΔHo = Σ products of ΔHof – Σ reactants of ΔHof
= [2 HNO3 + NO] – [3NO2 + H2O]
= [2(-207.4) + (90.2)] – [3(33.2) + (-285.8)]
= -138.4 kJ
14. A
ΔH = Σ (Bond energies of reactants) – Σ (Bond energies of products)
= [C3H8 + 5O2] – [3CO2 + 4H2O]
= [C-H×8 + C-C×2 + O=O×5] – [3(C=O×2) + 4(O-H×2)]
= [410×8 + 340×2 + 500×5] – [3(730×2) + 4(460×2)]
= 6460 – 8060
= -1600kJ
15. C
ΔS > 0 means the increasing disorder by the spontaneous reaction. I and IV are spontaneous reactions. II and III are non-spontaneous reactions.
16. A
The phase change from solid to gas has the greatest increasing entropy among the reactions shown.
17. A
When an endothermic processes, the temperature of its surroundings is decreased and the entropy is decreased.