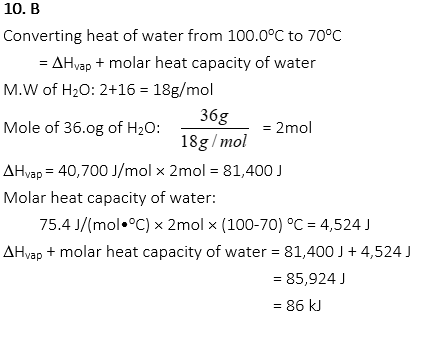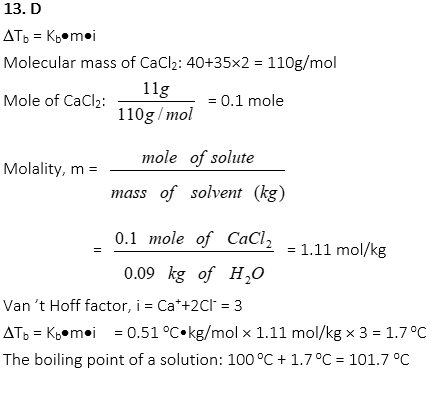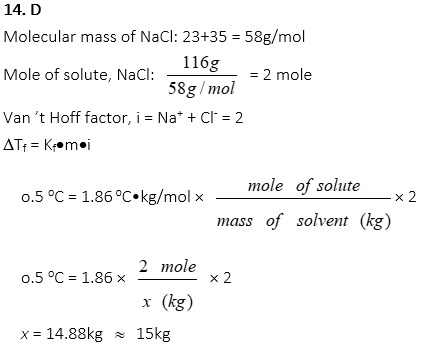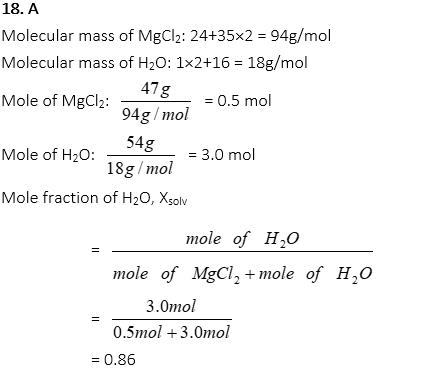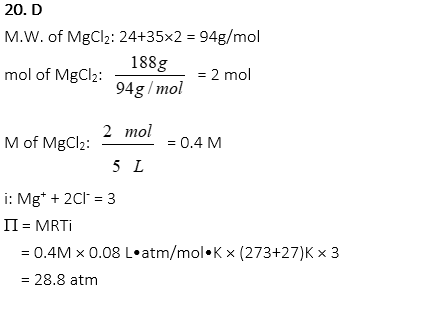VI. Phases of matter: Solid, Liquid, and Gas questions

Phase of matter and phase change problems
There are 22 phase of matter and phase change problems , which are important content-oriented in the summary part of this chapter. Click the ‘Answer’ button to see the corresponding answers and explanations, and click the ‘Go back’ button to slove the next questions.
1. Which of the following is NOT a feature of liquid?
A. No attraction force between molecular particles
B. Surface tension
C. Viscosity
D. Adhesion
E. Fluidity
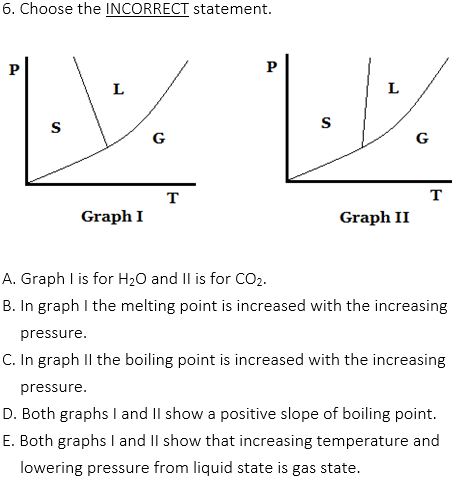
2. Which of the following is an INCORRECT statement?
A. High boiling point means low vapor pressure.
B. Boiling point is the temperature at which the vapor pressure of a liquid is equal to the external pressure of the atmosphere.
C. The sphere shape of drops in the air can be explained by surface tension.
D. Liquid has stronger attraction between molecular particles than solid.
E. The movement of individual atoms of gas is random.
3. Changing phase state from gas to solid is called _______________ and from gas to liquid is called
_______________.
A. Sublimation, fusion
B. Deposition, condensation
C. Freezing, vaporization
D. Sublimation, condensation
E. Vaporization, condensation
5. Iodine and dry ice absorb heat from the surroundings into the system and then change their phase from solid to gas. This is called _______________ .
A. Sublimation
B. Vaporization
C. Fusion
D. Freezing
E. Deposition
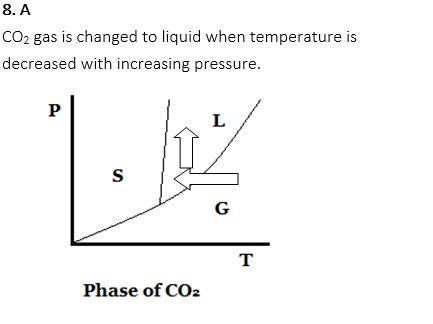
8. What should the beverage factory do to resolve CO2 gas into the carbonated beverage easily?
A. Decrease temperature and increase pressure
B. Increase temperature and decrease pressure
C. Decrease temperature and decrease pressure
D. Increase temperature and increase temperature
E. None of above
9. The triple point of a substance is all of the following EXCEPT ____________________.
A. Coexistence point of solid, liquid, and gas
B. Indistinguishable area between liquid and gas
C. The pressure of solid, liquid, and gas is in a state of equilibrium
D. The temperature of solid, liquid, and gas is in a state of equilibrium
E. All are correct.
10. How much heat energy will be released when the 36.0g of water at 100.0oC is cooled and the final temperature is 70oC? (The enthalpy of vaporization of water, DHvap: 40.7 kJ/mol, the molar heat capacity of water: 75.4 J/(mol•oC))
A. 45 kJ
B. 86 kJ
C. 172 kJ
D. 1547 kJ
E. 1655 kJ
13. What is the boiling point of a solution when 11.0g of CaCl2 is completely dissolved in 90.0g of water? (Kb of water = 0.51 oC•kg/mol)
A. 120.2 oC
B. 110.1 oC
C. 105.6 oC
D. 101. 7 oC
E. 98.3 oC
14. Approximately how many grams of water would be needed to decrease freezing point of the solution to -o.5 oC when 116g of NaCl is dissolved to the solution completely? (Kf of water = 1.86oC•kg/mol)
A. 8kg
B. 10kg
C. 13kg
D. 15kg
E. 18kg
15. Which of the following would have the lowest freezing point when cation (C) and/or anion (A) is in water? (Molar freezing point-depression constant, Kf, is equal.)
A. 0.3 molality with CA2
B. 0.2 molality with A2A5
C. 0.2 molality with C2A3
D. 0.5 molality with A3A5
E. 0.3 molality with C2A
16. 5.0g of unknown compound is added to 100.0g of camphor (C10H16O) and the normal boiling point of the solution is increased to 1.65 oC. Identify the unknown compound. (Unknown compound is non-electrolyte and Kb of camphor is 5.95 oC•kg/mol.)
A. C9H8O4
B. C6H6
C. CHCl3
D. C4H10O
E. C2H6O
17. Unlike fresh water, sea water seldom freezes during winter. All of the following are similar or related phenomena EXCEPT ____________________ .
A. Antifreeze in a vehicle
B. Lower freezing point of the solution than the pure solvent
C. Calcium chloride on the road
D. Colligative properties
E. All are related phenomena.
18. What is the mole fraction of water when 47.0 g of MgCl2 is completely dissolved in 54.0g of water?
A. o.86
B. 0.14
C. 0.33
D. 0.67
E. 0.28
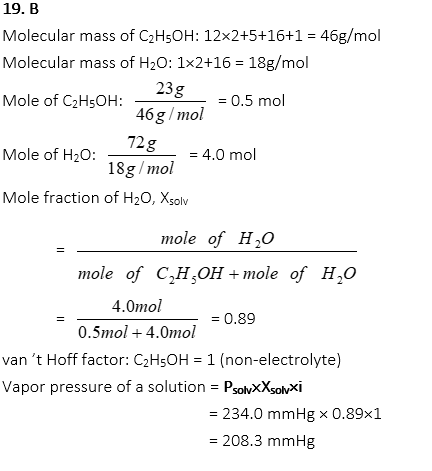
19. What is the vapor pressure of a solution when 23.0g of ethyl alcohol, C2H5OH, is completely dissolved in 72.0g of water at 70 oC. (The vapor pressure of water at 70 oC = 234.0 mmHg.)
A. 26 mmHg
B. 208 mmHg
C. 226 mmHg
D. 328 mmHg
E. 460 mmHg
20. A 5L solution is prepared with 188g of MgCl2 in water. What is the osmotic pressure at 27 oC? (R=0.08 L•atm/mol•K)
A. 0.9 atm
B. 2.6 atm
C. 9.6 atm
D. 28.8 atm
E. 37.6 atm
21. A mixture of sugar, C12H22O11, dissolved in 5L of solution gives 182.4 mmHg of osmotic pressure at 300K. What is the mole of the mixture of sugar? (R=0.1 L•atm/mol•K)
A. 0.04 mol
B. 0.08 mol
C. 0.4 mol
D. 0.8 m0l
E. 2.0 mol
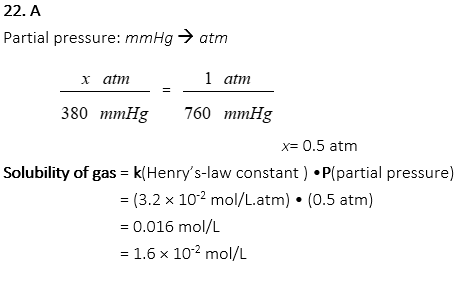
22. What is the solubility (mol/L) of CO2 in water under partial pressure of 380 mmHg at 25 oC if the Henry’s-law constant of CO2 is 3.2 × 10-2 mol/L.atm?
A. 1.6×10-2
B. 6.4×10-2
C. 8.4×10-2
D. 12.2
E. 15.6
Answers
1. A
Surface tension, viscosity, adhesion, and fluidity are the specific characteristics of liquid. Solid and liquid have the attraction force between molecular particles.
2. D
Solid has stronger attraction between its molecular particles than liquid. The order of strength of attraction between molecular particles: gas < liquid < solid
4. D
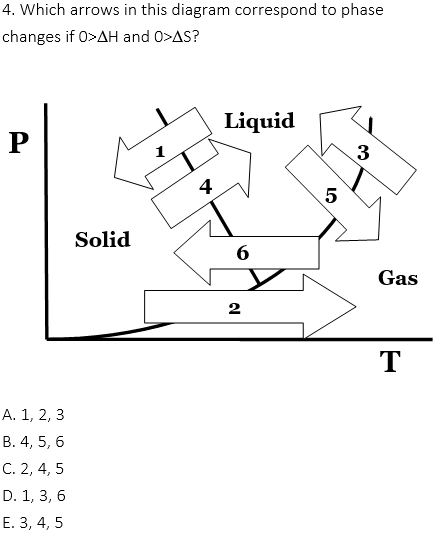
0>DH, 0>DS: condensation (3), freezing (1), deposition (6)
0<DH, 0<DS: vaporization (5), fusion (4), sublimation (2)
7. A
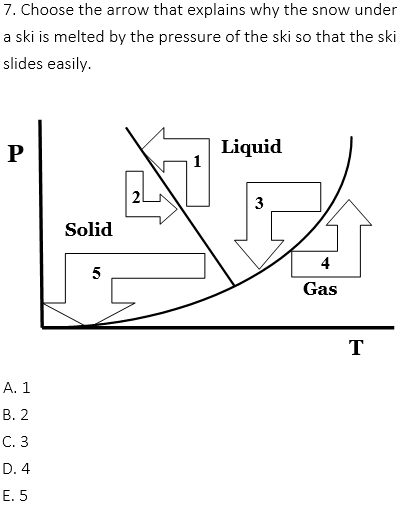
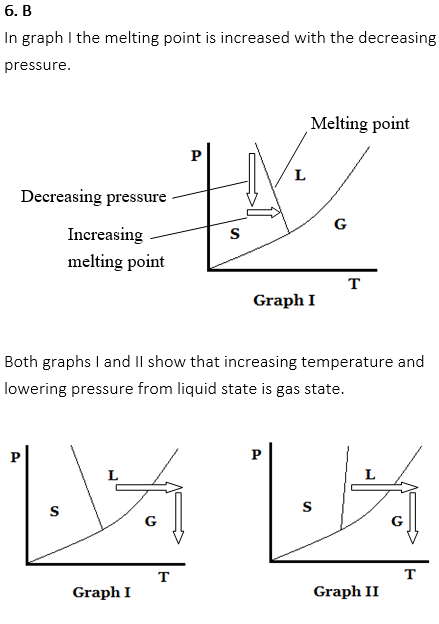
The melting point of snow (solid) is decreased by the increasing pressure of the ski. This is why the ski slides easily.
12. D
Because of the attraction forces between solute and pure solvent, the surface area of vaporizable solvent is decreased. (See the Fig. 5 Vapor pressure of pure solvent and solution.)
15. C
From DTf = Kf·m·i, the van ’t Hoff factor, i, means the mole of solute ions (cation + anion).
The freezing point becomes lower with the higher the van ’t Hoff factor, i.
0.3 molality with C2A = 0.3×(2+1) = 0.9
0.2 molality with A2A5 = 0.2×1 = 0.2
0.2 molality with C2A2 = 0.2×(2+3) = 1.0
0.5 molality with A3A5 = 0.5×1 = 0.5
0.3 molality with C2A = 0.3×(2+1) = 0.9
Therefore, the lowest freezing point is C.
17. E
All are similar/related. Sea water has a lower freezing point than fresh water because non-volatile solution (solvent + solute) has lower freezing point than pure non-volatile solvent. This is one of colligative properties. A, C, and D are the examples of the colligative properties.