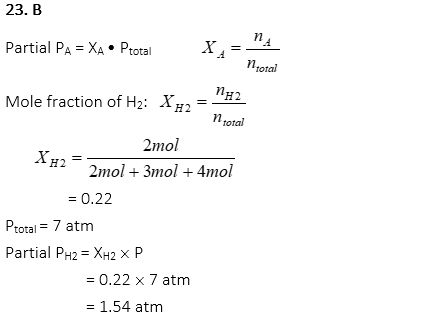VII. Gases

Gas law questions
There are a total of 28 gas law questions, which are important content-oriented in the summary part of this chapter. Click the ‘Answer’ button to see the corresponding answers and explanations, and click the ‘Go back’ button to slove the next questions.
1. The melting point of Manganese, Mn, which is used for industrial metal alloy or stainless steel, is 2,275oF. Convert this Fahrenheit reading to Kelvin.
A. 1,246 K
B. 1,519 K
C. 2,548 K
D. 4,127 K
E. 4,400 K
2. Which of the following does NOT apply to the Kinetic molecular theory of gases?
A. Van der Waals equation
B. No any force between gas molecules
C. No change of energy from elastic collision
D. Increasing Kinetic energy with increasing temperature
E. Random and straight movement
3. The volume of an ideal gas in a cylinder is 44.8L at 4 atm. What is the pressure of this gas if a piston compresses its volume to 22.4L?
A. 2 atm
B. 6 atm
C. 16 atm
D. 3,040 mmHg
E. 6,080 mmHg
4. A 4.4L of sample of air in a container is cooled from 27oC to 0oC. What is the final volume of this air?
A. 0.16L
B. 2.0L
C. 4.0L
D. 4.8L
E. 118.8L
5. The pressure of an unknown gas in a cylinder was 30 atm at 240K. What is the torr of this gas when the temperature of this gas is heated to 400K?
A. 50 torr
B. 144 torr
C. 7,200 torr
D. 38,000 torr
E. 109,440 torr
6. The initial temperature of an unknown gas is placed in a 2L container is 25oC at 3atm. If the condition of the gas is changed to 12atm and 1L of volume, what is the new temperature?
A. 50oC
B. 149K
C. 298K
D. 323oC
E. 596oC
7. The pressure of a gas in a balloon is 2 atm with a volume of 200L at 27 oC. What is the volume of the gas when the pressure of the balloon is changed to 4 atm at 47 oC?
A. 53.4 L
B. 57.4 L
C. 106.7 L
D. 114.8 L
E. 213.4 L
8. 6 mole of CO2 is contained in 3L of a cylinder at 2.0atm pressure. When 3 mole of this gas is contained in 6L of the cylinder with the same temperature, what is the pressure of this gas?
A. 0.1 atm
B. 0.2 atm
C. 0.5 atm
D. 1.0 atm
E. 2.0 atm
9. 96g of a gas is contained in a 73.8 L cylinder at 27oC. The pressure is 760 mmHg. What is the gas? (Gas constant, R = o.o82 atm•L/ mol•K).
A. He
B. Ne
C. H2
D. O2
E. CO2
10. Which of the following gases has the highest temperature at a constant pressure?
A. 40g of Ar
B. 2g of He
C. 22g of CO2
D. 56g of CO
E. 7g of Cl2
11. 2 mol of O2, 1 mol of CO2, and 3mol of N2 are contained in 20L of a flask at 0oC. What is the total pressure of this gas mixture?
A. 1.12 atm
B. 2.24 atm
C. 3.36 atm
D. 5.60 atm
E. 6.72 atm
12. When 32g of methane and 44g of carbon dioxide are contained in a 44.8L cylinder, the total pressure is 5 atm. What is the temperature of the gas mixture in this cylinder?
A. 911 K
B. 2732 K
C. 1366 K
D. 455 K
E. 683 K
13. If 34g of NH3 gas is placed in a 44.8L container at a pressure of 1000mmHg, what is the temperature of NH3?
A. 21.1K
B. 86.4oC
C. 254.3K
D. 15795oC
E. 273170.0K
14. What is the density (g/L) of 3.0 atm of CO2 at a room temperature of 27 oC? ( R= 1.0 atm•L/mol•K)
A. 8.8
B. 4.4
C. 2.2
D. 0.88
E. 0.44
15. The density of O2 is 4.3 g/L at 0oC. What is the pressure?
A. 1.0 atm
B. 2.0 atm
C. 3.0 atm
D. 4.0 atm
E. 6.0 atm
16. The density of unknown gas is 3.2g/L at STP. What is this gas?
A. Hydrogen
B. Neon
C. Fluorine
D. Chlorine
E. Methane
17. The density of 56g of an unknown gas at STP is 5.0 g/L. How many molecules of this gas are present?
A. 3.01×1023
B. 6.02×1023
C. 1.20×1024
D. 2.02×1024
E. 4.80×1025
19. Which of the following two gases has the highest ratio of diffusion?
A. H2 and He
B. H2 and Ar
C. CO2 and He
D. He and N2
E. H2 and O2
20. Find the molecular mass of an unknown gas that is 2 times faster than SO2.
A. 4 g/mol
B. 16 g/mol
C. 32 g/mol
D. 128 g/mol
E. 256 g/mol
22. When this stopcock is opened, what is the total pressure of the gas mixture from the above question?
A. 0.9 atm
B. 1.2 atm
C. 1.5 atm
D. 1.8 atm
E. 2.5 atm
23. A gas mixture of 2 mol of H2, 3mol of NH3, and 4 mol of O2 is contained in a flask at a total pressure of 7 atm. What is the partial pressure of H2?
A. 0.22 atm
B. 1.54 atm
C. 3.08 atm
D. 15.9 atm
E. 31.8 atm
24. A gas mixture of 28g of CO, 34g of NH3, and 10g of H2 is contained in a flask of 5L. What is the total pressure when the partial pressure of NH3 is 5 atm?
A. 1.25 atm
B. 2.36 atm
C. 10.6 atm
D. 20.0 atm
E. 40.0 atm
25. What is the mole fraction of N2 if a gas mixture has partial pressures of 3 atm of N2 and 5 atm of CO2?
A. 0.37
B. 0.63
C. 0.83
D. 1.60
E. 2.67
26. The chemical reaction below is used for an air bag in a car. When the car is bumped, the air bag, by producing N2 gas, inflates and saves the life of a driver. If the gas N2 is produced at a volume of 44.8L at STP, how many grams of NaN3 are used for the air bag?
2NaN3(s) → 2Na(s) + 3N2(g)
A. 43.3 g
B. 86.5 g
C. 129.9 g
D. 173.0 g
E. 259.5 g
27. Hg and O2 are produced by the decomposition of 651g of HgO at 1.2 atm and 25oC. How many liters of O2 are produced?
2HgO(g) → 2Hg(l) + O2(g)
A. 2.6 L
B. 20.4 L
C. 22.4 L
D. 30.5 L
E. 40.7 L
28. When 56g of N2 is reacted with an excess of H2, the volume of 44.8L of NH3 is produced at 0oC. What is the pressure of NH3?
N2(g) + 3H2(g) → 2NH3(g)
A. 0.5 atm
B. 1.0 atm
C. 2.0 atm
D. 2.5 atm
E. 4.0 atm
Answers
2. A
The kinetic molecular theory of ideal gas can be summarized as “No forces between gas molecules, random and straight movement, negligible size, and elastic collisions.” Van der Waals equation predicts the interaction of molecules of real gases.