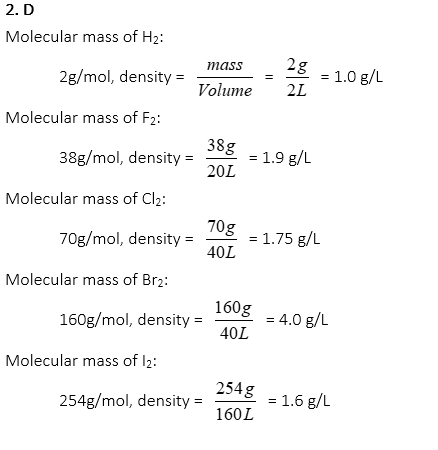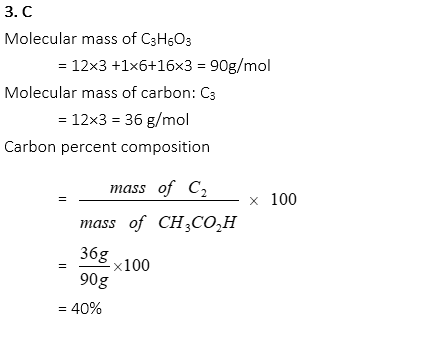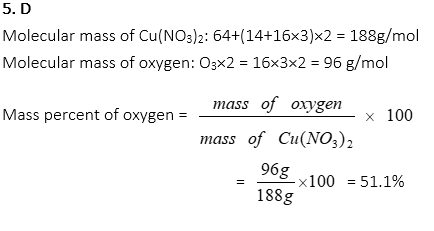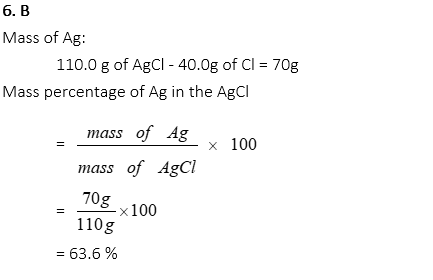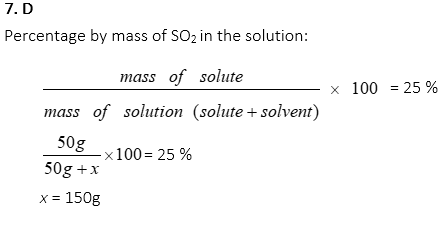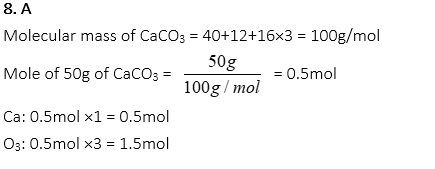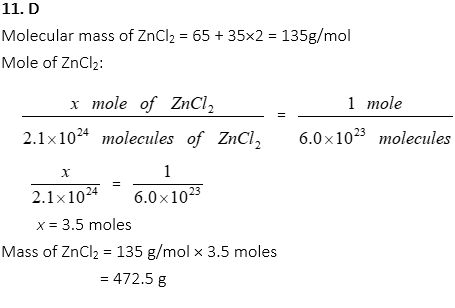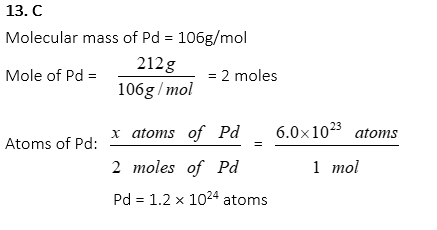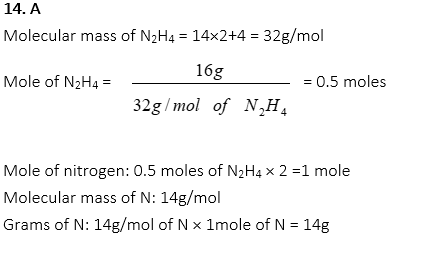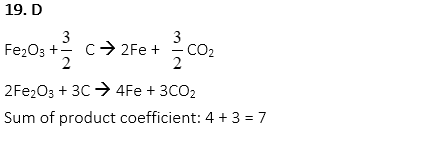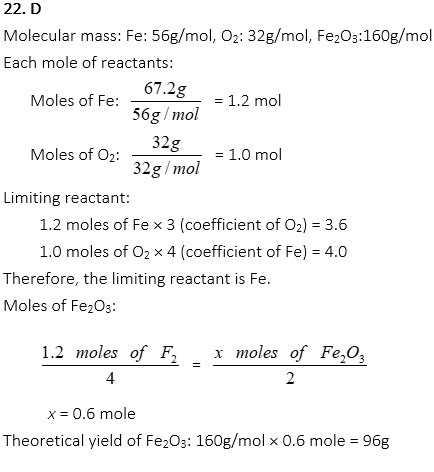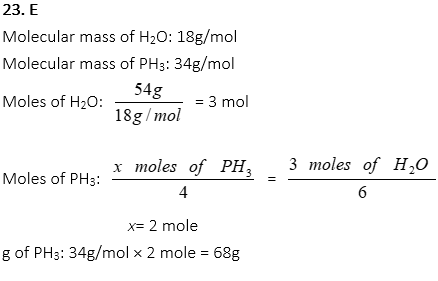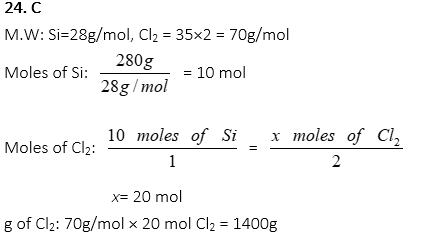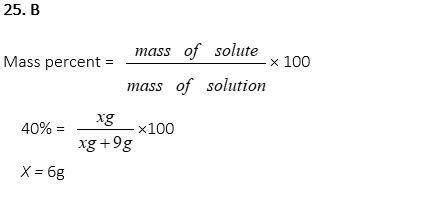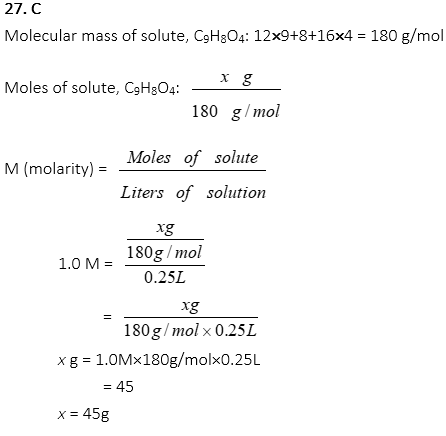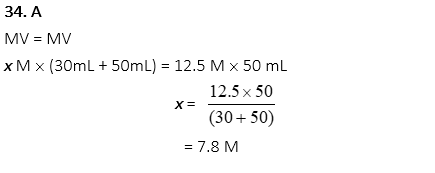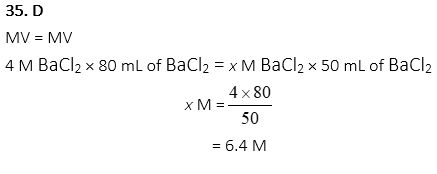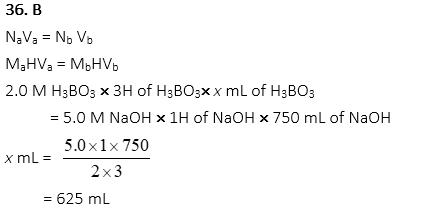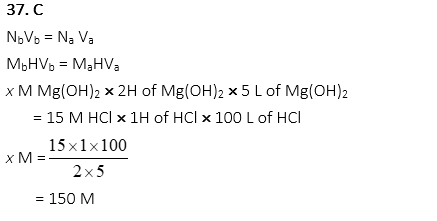IV. Stoichiometry and Solution questions

Stoichiometry practice problems
There are a total of 48 stoichiometry practice problems, which are important content-oriented in the summary part of this chapter. Click the ‘Answer’ button to see the corresponding answers and explanations, and click the ‘Go back’ button to slove the next questions.
1. What is the density when 32g of an unknown compound is contained in the 2cm edge of a cube?
A. 32 g/cm3
B. 16 g/cm3
C. 8 g cm3
D. 4 g/cm3
E. 2 g/cm3
2. Which of the following has the highest density at STP?
A. H2 in 2L container
B. F2 in 20L container
C. Cl2 in 40L container
D. Br2 in 40L container
E. I2 in 160L container
5. What is the mass percentage of oxygen in Cu(NO3)2, approximately?
A. 9 %
B. 26 %
C. 43 %
D. 51 %
E. 65 %
6. 40 g of Cl is found in 110 g of AgCl compound. What is the mass percentage of Ag in the compound?
A. 36.4%
B. 63.6%
C. 107.9%
D. 98.1%
E. 67.8%
7. 50g of SO2 is to be dissolved in water. If the mass percentage of SO2 in the solution is 25%, then how many grams of water should be prepared?
A. 12.5g
B. 50.0g
C. 100.0g
D. 150.0g
E. 200.0g
8. How many moles of calcium and oxygen each are found in 50g of CaCO3?
A. 0.5mole of calcium, 1.5mole of oxygen
B. 0.5mole of calcium, 0.5mole of oxygen
C. 1.5mole of calcium, 1.5mole of oxygen
D. 1.2mole of calcium, 1.0mole of oxygen
E. 1.2mole of calcium, 3.6mole of oxygen
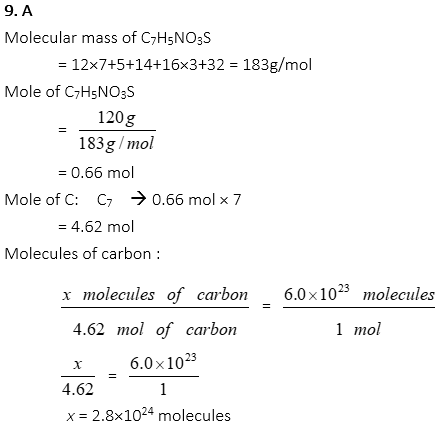
9. How many molecules of carbon are in 120g of saccharin, C7H5NO3S, approximately?
A. 2.8×1024
B. 4.0×1023
C. 6.0×1023
D. 2.0×1023
E. 1.2×1024
10. How many grams of glucose, C6H12O6, are equal to 2 moles of glucose?
A. 90g
B. 180g
C. 360g
D. 400g
E. 460g
11. If ZnCl2 contains 2.1×1024 atoms, what is the mass of zinc chloride?
A. 39 g
B. 135 g
C. 270 g
D. 473 g
E. 2.8 × 1026 g
12. If sodium oxide, Na2O, contains 12 × 1022 atoms, which of the following is an INCORRECT statement?
A. The mass of Na2O is 12.4 g.
B. The atoms of cation is 2.4×1023.
C. The total moles of ions is 0.2.
D. The mass of O is 3.2g.
E. The atomic numbers of sodium oxide is equal to anion.
13. Palladium, Pd, is one of the important metals for making PFM (porcelain fused metal) crowns in a dental laboratory. How many atoms are in 212g of Pd?
A. 3.0 × 1023
B. 6.0 × 1023
C. 1.2 × 1024
D. 6.4 × 1024
E. 1.2 × 1025
15. Unknown compound is composed of 224g of iron and 96g of oxygen. What is the empirical formula for the unknown compound?
A. FeO3
B. Fe2O6
C. Fe2O3
D. Fe4O6
E. Fe3O
16. Percent composition of an unknown compound is 43.4% Na, 11.3% C, and 45.3% O by mass. What is the empirical formula for the unknown molecule?
A. Na2CO3
B. Na4CO6
C. NaC2O3
D. Na8C2O12
E. Na2CO6
17. The empirical formula of Caproic acid is C3H6O. What is the molecular formula when the molecular weight of Caproic acid is 116 amu?
A. C1H2O0.5
B. C3H6O
C. C6H12O2
D. C9H18O3
E. C12H24O4
18. The molecular mass of an unknown compound is 58g/mol. Which of the following could be the empirical formula for the unknown compound?
A. C4H10
B. C2H5
C. C2H6
D. CH3
E. C2H3
19. Carbon is reacted with Fe2O3 to produce iron. What is the sum of the product coefficient when the following equation is balanced?
Fe2O3 + C → Fe + CO2
A. 2
B. 3
C. 5
D. 7
E. 9
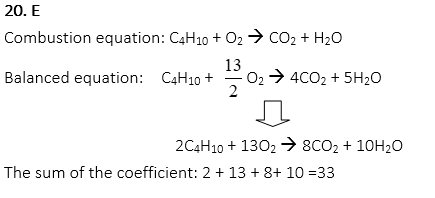
20. Butane is burned in air. What is the sum of the coefficient from the balanced equation for the reaction?
A. 4
B. 15
C. 18
D. 25
E. 33
21. 180g of C3H8O reacted with an excess of O2 produces 277.2g of CO2. What is the percent yield of CO2?
2C3H8O + 9O2 → 6CO2 + 8H2O
A. 70%
B. 75%
C. 80%
D. 85%
E. 90%
22. If 67.2g of Fe reacts with 32.0g of O2, how many grams of Fe2O3 are produced from the following equation?
4Fe + 3O2 → 2Fe2O3
A. 43g
B. 52g
C. 80g
D. 96g
E. 172g
23. How many grams of PH3 are used to produce 54g of H2O in the following reaction? (O2 is the excess reactant)
4PH3 + 8 O2 → P4O10 + 6H2O
A. 17g
B. 34g
C. 51g
D. 60g
E. 68g
24. How many grams of Cl2 are required to react with 280g of Si in the following reaction?
Si + 2Cl2 → SiCl4
A. 350g
B. 700g
C. 1400g
D. 2800g
E. 3500g
25. How many g of MgCl2 is dissolved in 9g of water when the mass percent of MgCl2 in the solution is 40%?
A. 3g
B. 6g
C. 9g
D. 12g
E. 15g
26. 400 mL of solution is created by dissolving 148 g of KCl in water. What is the molarity of the KCl solution?
A. 5 M
B. 10 M
C. 20 M
D. 185 M
E. 370 M
27. How many g of aspirin, C9H8O4, are prepared in 250mL of dichloromethane, CH2Cl2, to produce a 1.o M solution of aspirin?
A. 0.25 g
B. 25 g
C. 45 g
D. 90 g
E. 180 g
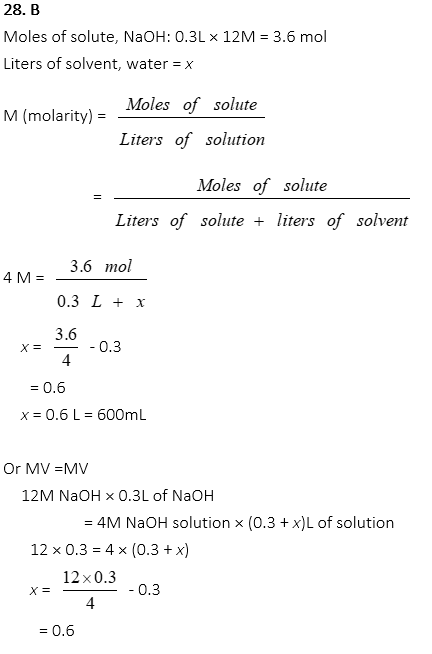
28. 300mL of 12M NaOH is dissolved in water to produce 4M NaOH solution. How much water is added?
A. 300 mL
B. 600 mL
C. 900 mL
D. 1200 mL
E. 1500 mL
29. What is the molar concentration of oxygen in a 0.25M H3PO4 solution?
A. 0.25 M
B. 0.5 M
C. 0.75 M
D. 1.0 M
E. 4.0 M
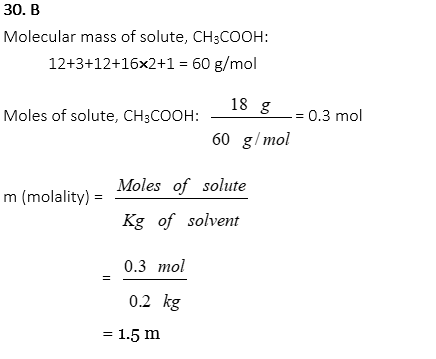
30. 18g of acetic acid, CH3COOH, is dissolved in 200g of water. What is the molality of the acetic acid solution?
A. 0.3 m
B. 1.5 m
C. 3.0 m
D. 4.5 m
E. 5.0 m
31. 400g of unknown compound is dissolved in 5kg of water. If the molality of the solution is 4m, which of the following is the unknown compound?
A. HF
B. NaOH
C. Ca(OH)2
D. H2SO4
E. HI
32. When 5.0M NaCl solution has a density of 1.09g/mL, what is the molality of this solution? (M.W. of NaCl = 58g/mol)
A. 3.50
B. 6.25
C. 10.40
D. 11.25
E. 15.50
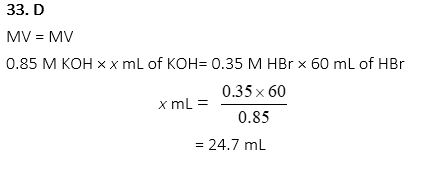
33. What volume of o.85M KOH(aq) is needed to neutralize 60mL of 0.35 M HBr?
A. 145.7 mL
B. 72.8 mL
C. 49.4 mL
D. 24.7 mL
E. 12.4 mL
34. What is the molarity of a final solution of KOH when 30.0mL of water is added to 50.0mL of 12.5M KOH?
A. 7.8 M
B. 15.6 M
C. 20.8 M
D. 31.2 M
E. 41.6 M
35. What is the final concentration of BaCl2 when 80ml of 4M BaCl2 solution is diluted to 50ml of BaCl2 solution?
A. 1.3M
B. 2.5M
C. 5.0M
D. 6.4M
E. 12.8M
36. When 2.0M boric acid, H3BO3, neutralizes 750 mL of 5.0M sodium hydroxide, NaOH, what volume of boric acid is needed?
A. 312 mL
B. 625 mL
C. 938 mL
D. 1250 mL
E. 1875 mL
37. What molarity of Mg(OH)2 is required when 5 L of Mg(OH)2 neutralizes 100 L of 15M HCl?
A. 1 M
B. 75 M
C. 150 M
D. 300 M
E. 600 M
38. What is the product from the reactant?
Pb(NO3)2(aq) + Na2SO4(aq) →
A. PbNa2(aq) + (NO3)2SO4(aq)
B. PbSO4(s) + 2NaNO3(aq)
C. PbNa2(aq) + 2NaNO3(aq)
D. PbSO4(s) + (NO3)2SO4(aq)
E. Pb(NO2)2(aq) + Na2SO3(aq) + O2(g)
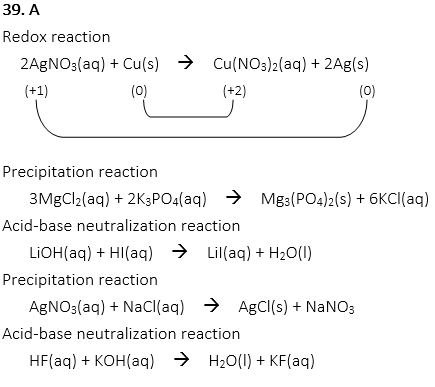
39. Which of the following is the redox reaction?
A. 2AgNO3(aq) + Cu(s) → Cu(NO3)2(aq) + 2Ag(s)
B. 3MgCl2(aq) + 2K3PO4(aq) → Mg3(PO4)2(s) + 6KCl(aq)
C. LiOH(aq) + HI(aq) → LiI(aq) + H2O(l)
D. AgNO3(aq) + NaCl(aq) → AgCl(s) + NaNO3
E. HF(aq) + KOH(aq) → H2O(l) + KF(aq)
41. Which of the following is NOT dissociated in water?
A. CH3COOH
B. KOH
C. C12H22O11
D. HNO3
E. KBr
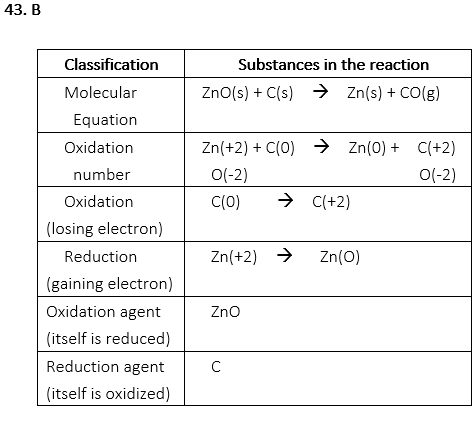
43. Which of the following is the reducing agent in the reaction below?
ZnO(s) + C(s) → Zn(s) + CO(g)
A. ZnO
B. C
C. Zn
D. CO
E. O
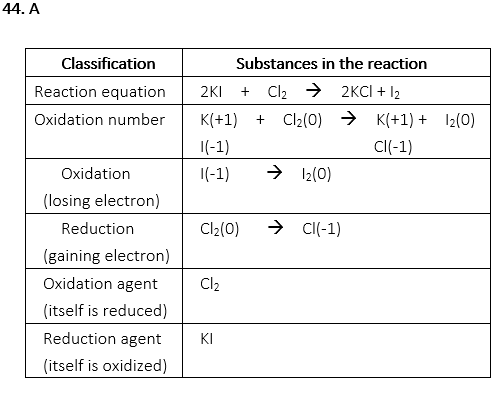
44. Which of the following is correct for the following reaction?
2KI + Cl2 → 2KCl + I2
A. Cl2 is the oxidizing agent.
B. KCl is the reducing agent.
C. Cl is oxidized.
D. I is reduced.
E. A and B
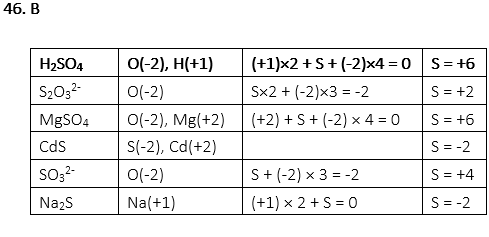
46. Which of the following compounds has the same oxidation number of S as in H2SO4?
A. S2O32-
B. MgSO4
C. CdS
D. SO32-
E. Na2S
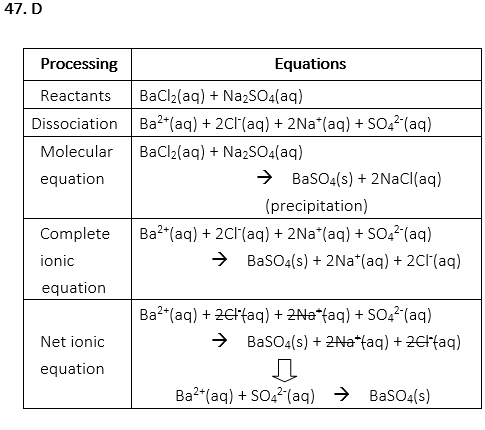
47. What is the net ionic equation from the following reactants?
BaCl2(aq) + Na2SO4(aq) →
A. Ba2+(aq) + 2Cl–(aq) + 2Na+(aq) + SO42-(aq)
B. 2Cl–(aq) + 2Na+(aq) → 2NaCl(aq)
C. BaCl2(aq) + Na2SO4(aq) → BaSO4(s) + 2NaCl(aq)
D. Ba2+(aq) + SO42-(aq) → BaSO4(s)
E. Ba2+(aq) + 2Cl–(aq) + 2Na+(aq) + SO42-(aq) → BaSO4(s) + 2Na+(aq) + 2Cl–(aq)
48. How many moles of water and chloride ion are needed to c0mplete the balanced redox reaction from the unbalanced net ionic equation?
Cr2O72- + Cl– → Cr3+ + Cl2
A. 2 moles of water, 12 moles of chloride ion
B. 7 moles of water, 14 moles of chloride ion
C. 7 moles of water, 6 moles of chloride ion
D. 1 moles of water, 6 moles of chloride ion
E. 14 moles of water, 12 moles of chloride ion
Answers
10. C
Molecular mass of C6H12O6 = 12×6+1×12+16×6 = 180g/mol
Grams of C6H12O6 = 180g/mol × 2 mol = 360g
17. C
Molecular mass of C3H6O: 12×3+6+16 = 58g/mol
Unknown constant x: x × 58g/mol = 116 g/mol
x = 2
Molecular formula: 2× C3H6O = C6H12O2
38. B
Pb(NO3)2(aq) + Na2SO4(aq)
Ion equation: Pb2+(aq) + 2NO3–(aq) + 2Na+(aq) + SO42-(aq)
Products: → PbSO4(s) +2NaNO3(aq)
40. A
When a halide (Cl–, Br–, I–) is bonded with Ag+, Hg2+, or Pb2+, it becomes an insoluble compound.
41. C
Alcohols (CH3OH, CH3CH2OH) and organic compounds (C12H22O11) are non-electrolytes in water.
42. D
Strong acids: HCl, HBr, HI, H2SO4, HNO3, HClO4
Weak acids: H3PO4, CH3COOH, HF, HCN
Strong bases: LiOH, NaOH, KOH, Mg(OH)2, Ca(OH)2, Sr(OH)2, KBr, NaCl, NaOH, ionic compounds
Weak bases: NH3, CH3NH2
48. C
Completed balanced redox reaction: Cr2O72- + 14H+ + 6Cl– → 2Cr3+ + 7H2O + 3Cl2
Processing | Equations |
Net ionic equation | |
Separation into oxidation and reduction | |
Balance atoms except H, O | |
Balance O by adding H2O | |
Balance H by adding H+ | |
Balance charges by adding electrons | 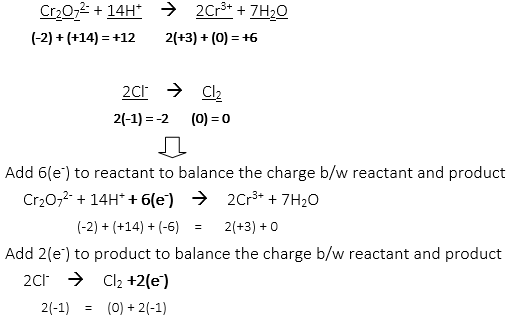 |
Multiply by coefficient to cancel electrons | 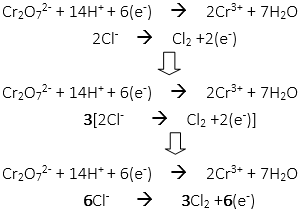 |
Cancel electrons and complete the balanced redox reaction | 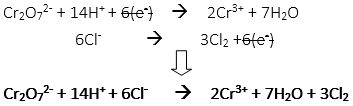 |