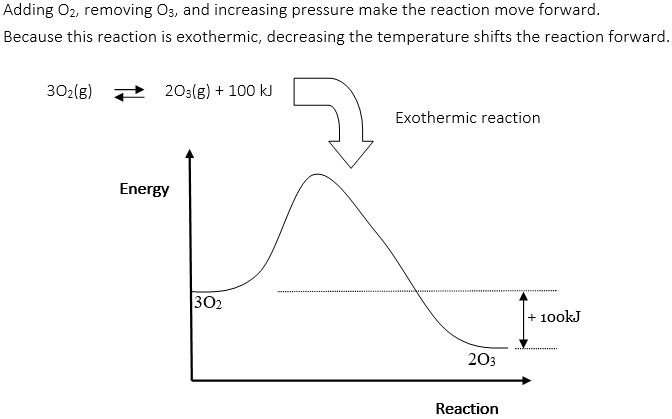
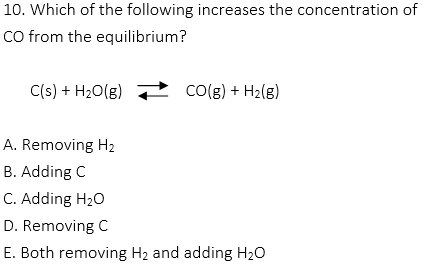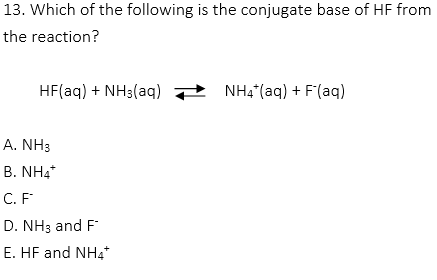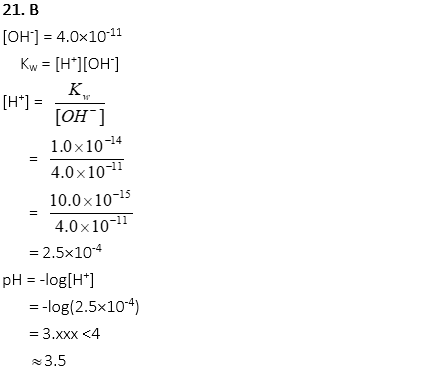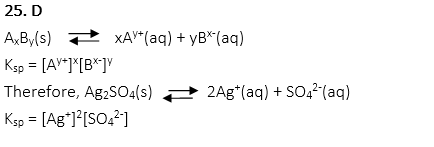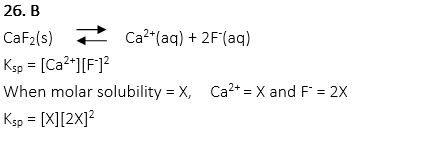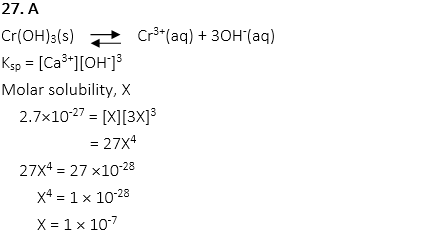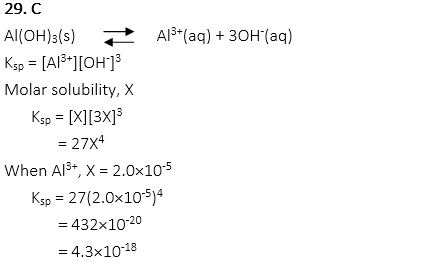IX. Chemical Equilibrium

Chemical equilibrium practice problems
There are a total of 30 chemical equilibrium practice problems, which are important content-oriented in the summary part of this chapter. Click the ‘Answer’ button to see the corresponding answers and explanations, and click the ‘Go back’ button to slove the next questions.
1. Which of the following is a reversible reaction?
A. 2C2H6(g) + 7O2(g) → 4CO2(g) + 6H2O(l)
B. 4HNO3(aq) + Cu(s) → Cu(NO3)2(aq) + 2NO2(g) + 2H2O(l)
C. LiOH(aq) + HI(aq) → Li(aq) + H2O(l)
D. Pb(NO3)2(aq) + 2KI(aq) → 2KNO3(aq) + PBI2(s)
E. CH4(g) + H2O(g) → CO(g) + 3H2(g)
2. Which of the following is a correct statement about the equilibrium state of a reaction?
A. The equilibrium state is seen in both reversible and irreversible reactions.
B. At equilibrium, the rate of a forward reaction is faster than in a reverse reaction.
C. At equilibrium, both the forward and reverse reactions are stopped.
D. At equilibrium, the concentration of the reactant and the product is constant.
E. At equilibrium, the concentration of the reactant and the product is the same.
3. Which of the following affects the equilibrium constant Keq?
A. Concentration of reactants or products
B. Temperature
C. Catalysis
D. Reaction quotient (Q)
E. B and C
4. Which of the following is an INCORRECT statement?
A. Keq > 1 means that there are more products than reactants.
B. Solids and pure liquids are not considered for the equilibrium equation.
C. Keq < 1 means that the forward reaction has not occurred.
D. Keq = Q represents the equilibrium state of a reaction.
E. The equilibrium depends on ΔG.
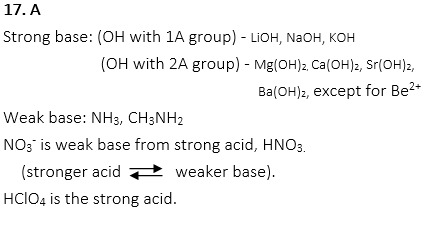
15. Which of the following CANNOT be a Bronsted-Lowry base?
A. H2PO4–
B. NH3
C. Cl–
D. H2O
E. BeCl2
16. Which of the following is a correct comparison of acid strength?
A. H2O > HF
B. HCl > HBr
C. HOCl > HOBr
D. HOClO2 > HOClO3
E. CH3CO2H > H2SO4
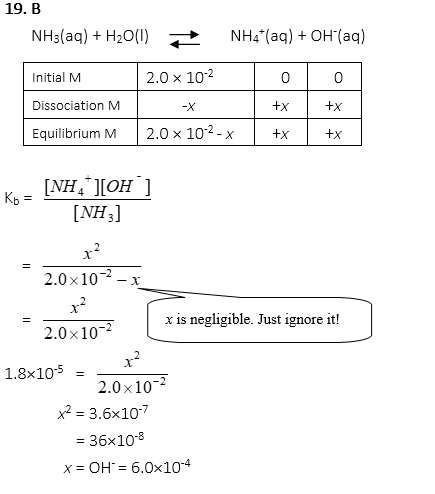
19. 0.02M NH3 is dissociated in a solution. When the Kb for NH3 is 1.8×10-5, what is the concentration of OH–?
A. 3.0×10-4 M
B. 6.0×10-4 M
C. 1.2×10-3 M
D. 1.8×10-3 M
E. 3.6×10-3 M
20. 0.05M H2SO4 is dissociated in a solution. What is the concentration of OH–?
A. 1.0×10-12
B. 2.0×10-13
C. 1.0 × 10-14
D. 3.0×10-15
E. 5.0×10-16
22. 48g of LiOH is dissolved in 4L of solution. What is the pH of the solution?
A. 0.5
B. 4.5
C. 9.0
D. 11.5
E. 13.5
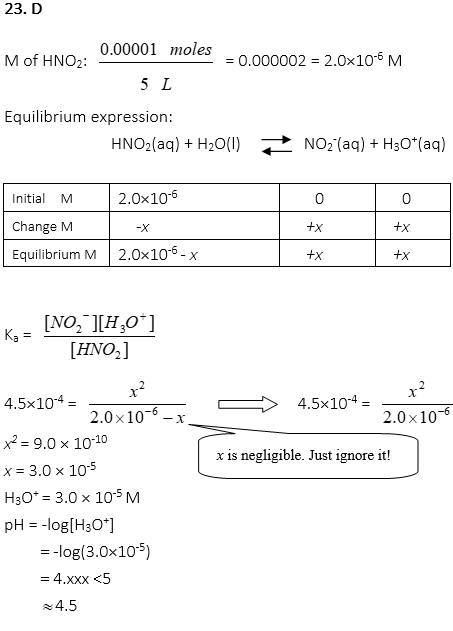
23. Ka of HNO2 is 4.5×10-4. When 0.00001 moles of HNO2 solution is diluted in a 5L vessel, find the pH of the solution.
A. 3.0
B. 3.5
C. 4.0
D. 4.5
E. 5.0
24. In a buffer solution a weak acid is 99% dissociated. When the pH is 6.5, what is the value of pKa?
A. 4.0
B. 4.5
C. 5.0
D. 5.5
E. 6.0
26. When the molar solubility is expressed by X, choose the correct equation of the solubility product Ksp for saturated solution of CaF2.
A. Ksp = [X][X]2
B. Ksp = [X][2X]2
C. Ksp = [X]2[2X]
D. Ksp = [2X][X]
E. Ksp = [2X]2[2X]
27. What is the molar solubility of Chromium(III) hydroxide, Cr(OH)3, when Ksp of Cr(OH)3 is 2.7×10-27 at 20oC.
A. 1 × 10-7 M
B. 3 × 10-7 M
C. 6 × 10-6 M
D. 1 × 10-5 M
E. 3 × 10-5 M
28. What is the concentration of Cu2+ in Cu3(PO4)2 when Ksp of the saturation solution Cu3(PO4)2 is 1.08×10-28?
A. 3×10-5 M
B. 1×10-6 M
C. 2×10-6 M
D. 3×10-6 M
E. 2×10-7 M
29. What is the Ksp for saturated solution Al(OH)3 when the concentration of aluminum ions is 2.0 × 10-5 M?
A. 4.0× 10-10
B. 2.7 × 10-18
C. 4.3×10-18
D. 8.6 × 10-16
E. 2.0 × 10-20
30. Which of the following is an INCORRECT statement?
A. Kc > 1 means that there is a predominance of product in the reaction.
B. Kc < Q means that the reaction moves from right to left.
C. IP < Ksp means that precipitation occurs in an ionic solution.
D. Decreasing Ksp means increasing the freezing point.
E. Increasing Ksp means increasing the electrical conduction.
Answers
1. E
A, B, C, and D are irreversible reactions, which are the combustion reaction, gas production from the reaction between an acid and a metal, neutralization between a strong acid and a strong base, and precipitation reaction.
2. D
The equilibrium state is seen only in a reversible reaction and the rate of a forward and reverse reaction is the same. The forward and reverse reaction does not stop but the reaction rate remains constant. The concentration of the reactant and the product is not the same, but the concentration of the reactant and the product is constant.
4. C
Keq < 1 means that the forward reaction is predominant and both forward and reverse reaction have occurred.
13. C
Conjugate acid-base pairs are formed by transferring one proton. The conjugate base from the acid HF is F–.
14. E
The reaction I is processed from right to left because HBr is a strong acid. F– is a Lewis base, which can donate an electron-pair. HF and BF3 are Lewis acids, which can receive an electron-pair. An Arrhenius base could be seen in a reaction that produces OH– in H2O. The concepts of Lewis acid and base involve the concepts of Bronsted-Lowry and Arrhenius.
15. E
A Bronsted-Lowry base is a substance that can receive H+.
H2PO4–, NH3, Cl–, and BeCl2 can receive H+ to be H3PO4, NH4+, HCl, H3O+.
However, BeCl2 cannot receive H+.
16. C
H2O < HF : binding with hydrogen, CH4 < NH3 < H2O < HF
HCl < HBr : binding with hydrogen, HF < HCl < HBr < HI
HOCl > HOBr : binding with hydro oxygen, HOI < HOBr < HOCl
HOClO2 < HOClO3 : binding with oxygen numbers, HOCl < HOClO < HOClO2 < HOClO3
CH3CO2H < H2SO4 : strong acid (HCl, HBr, HI, HClO4, H2SO4, HNO3), weak acid (H3PO4, CH3CO2H, HF, HCN)